Red blood cells serve as important glucose sink in high altitude-induced glucose tolerance
Published:
“ ” —
” —
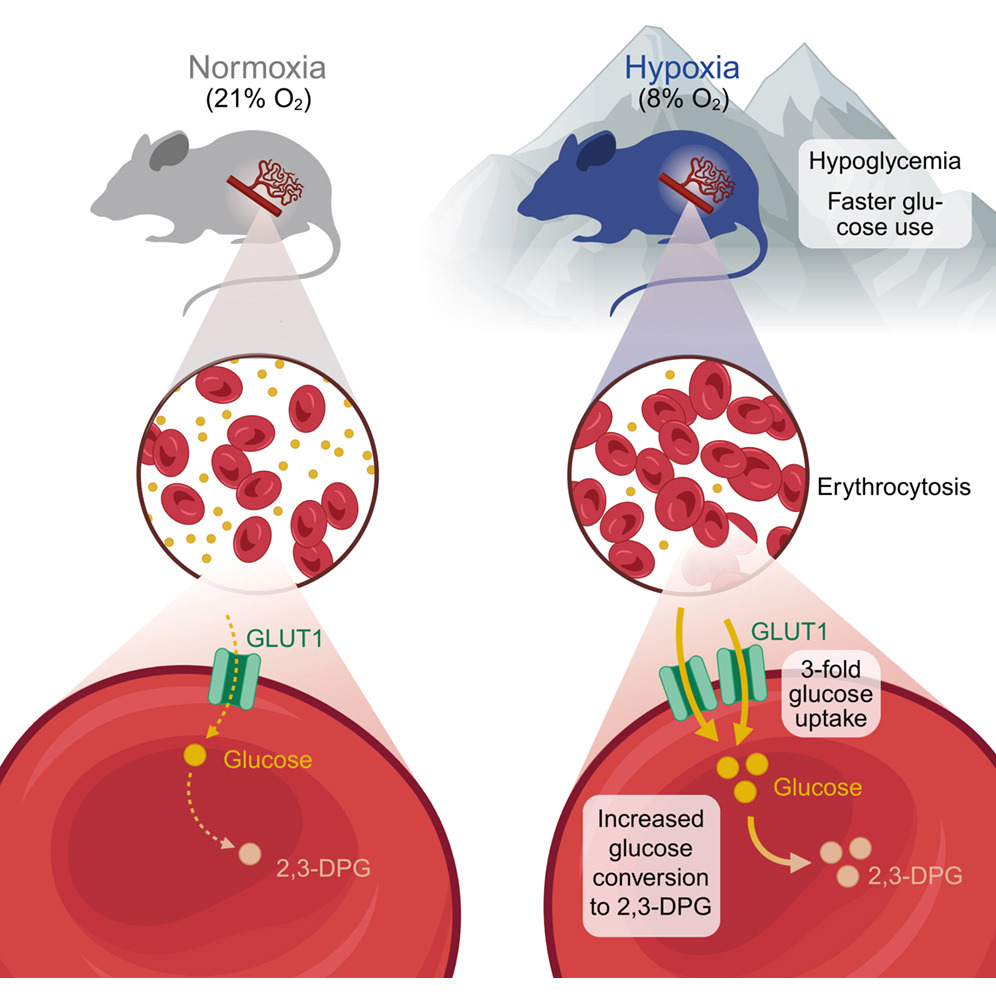
Epidemiological studies have reported an inverse correlation between living at high altitude and the prevalence of diabetes. High-altitude environments expose residents to multiple environmental stressors, including reduced oxygen availability, increased ultraviolet (UV) radiation, and lower temperatures. However, the mechanisms underlying the lower incidence of diabetes among high-altitude populations have remained unclear.
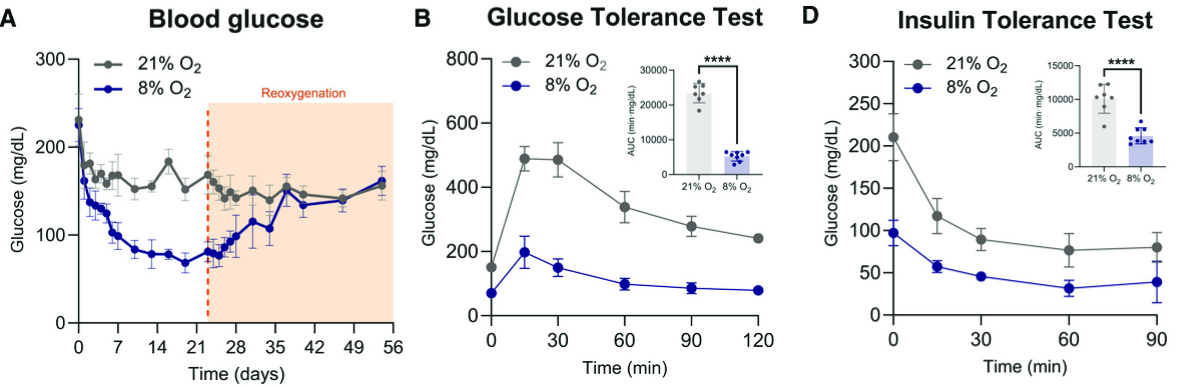
In a recent study published in Cell Metabolism (Marti-Mateos et al., 2026., Cell Metabolism), a research group led by Isha Jain at the University of California, San Francisco (UCSF) uncovered a potential explanation. The team demonstrated that erythrocytes function as a glucose “shunt” during hypoxia-induced hypoglycemia. Using C57BL/6J mice housed under normoxic (21% O₂) or hypoxic (8% O₂) conditions for 14 days, the researchers observed significantly lower blood glucose levels in mice exposed to hypoxia (Fig. 1A). Moreover, hypoxic mice displayed improved glucose tolerance, and this metabolic benefit persisted for months after the animals were returned to normoxic conditions (Fig. 1B). Notably, this phenotype did not depend on enhanced insulin signaling but instead appeared to result from a systemic physiological adaptation.
“ ” —
” —
Fig. 1 Shows evidence of hypoxia-induced hypoglycemia in animal model —
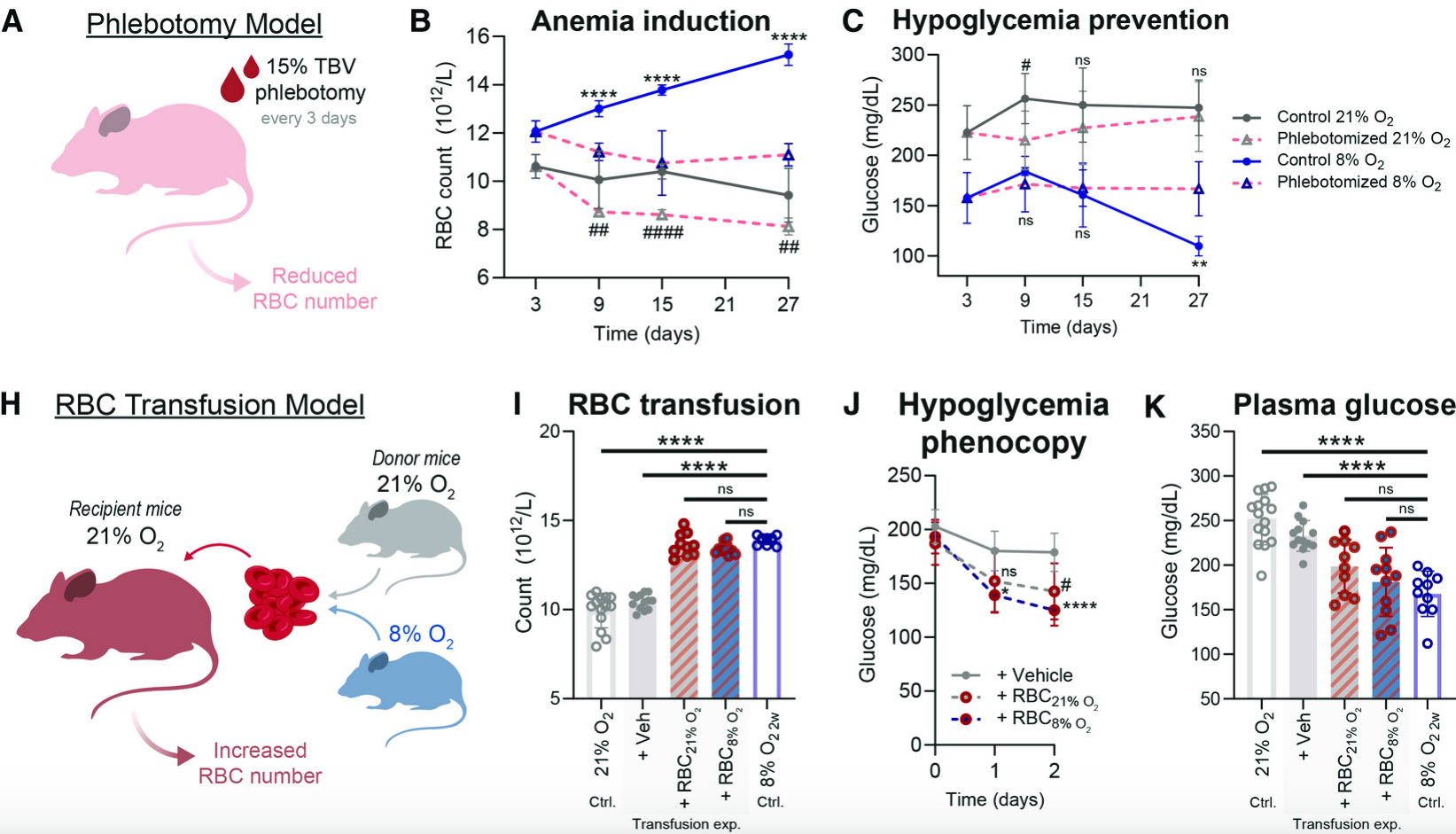
Erythrocytes are the most abundant cells in the body and rely primarily on the pentose phosphate pathway (PPP) and the Embden–Meyerhof–Parnas (EMP) glycolytic pathway due to their lack of mitochondria. Because hypoxic exposure increases both erythrocyte numbers and their glycolytic activity, the researchers hypothesized that erythrocytes may act as a glucose sink during hypoxia. To test this, the team performed serial phlebotomy every three days to induce anemia—thereby reducing erythrocyte numbers—in both normoxic and hypoxic mice. By withdrawing up to 15% of total blood volume (TBV), they asked whether decreasing erythrocyte abundance would attenuate the hypoxia-induced reduction in blood glucose levels. Indeed, repetitive blood withdrawal blunted the hypoglycemic effect and reversed the improved glucose tolerance observed in hypoxia-exposed mice. Importantly, this intervention did not significantly alter other hematological parameters aside from erythrocyte counts, supporting the idea that increased erythrocyte abundance directly contributes to systemic glucose lowering.
“ ” —
” —
Fig. 2 Shows the direct proof of hypoxia-induced erythrocytosis in promoting blood glucose reduction —
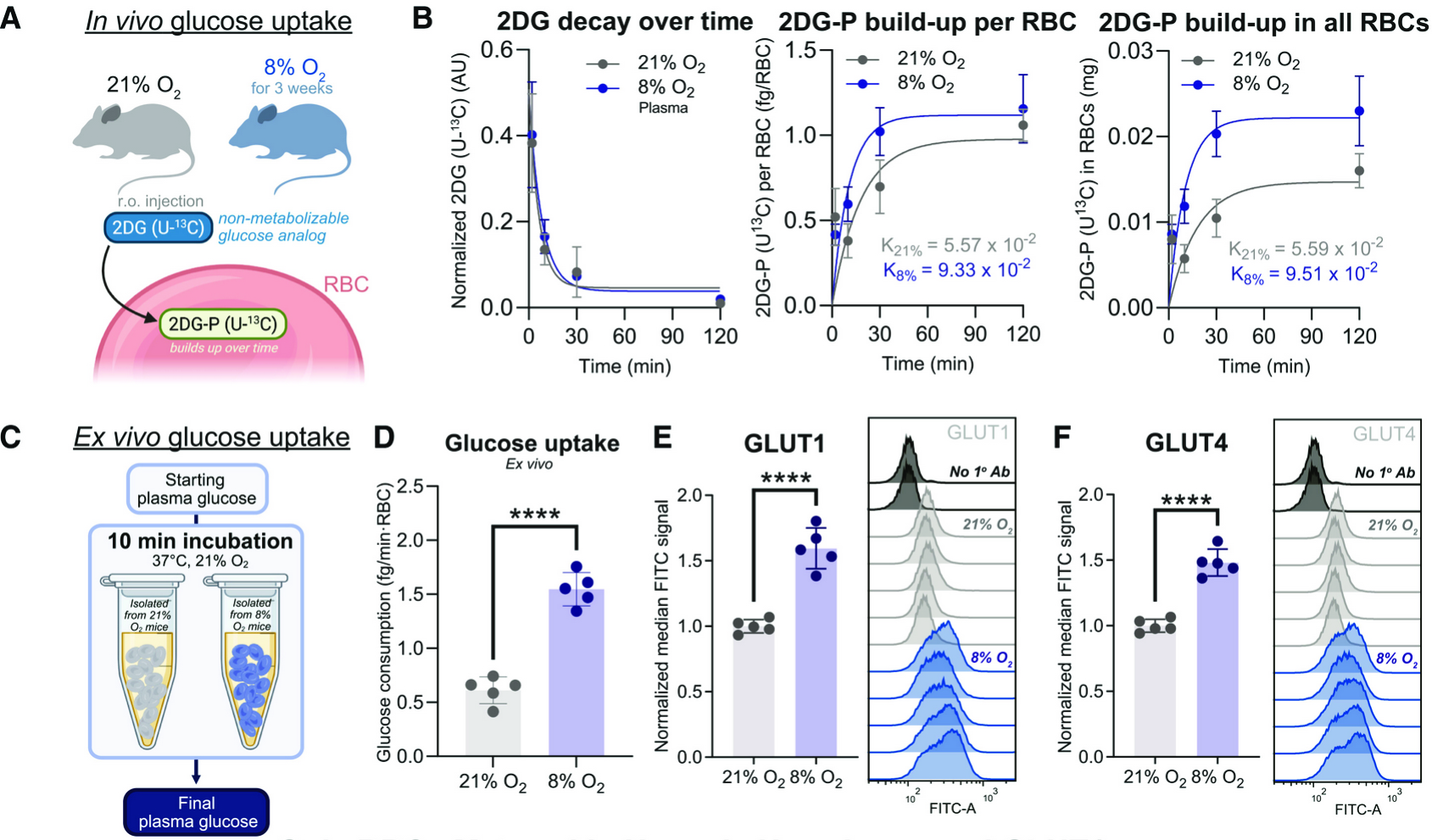
Next, the team performed an elegant red blood cell (RBC) transfusion experiment, transferring erythrocytes isolated from either hypoxic- or normoxic-housed mice into recipient animals to further dissect the causal role of elevated erythrocytes in mediating this metabolic adaptation. Interestingly, both transfusion groups exhibited hypoglycemia, with a slightly greater effect observed in mice receiving hypoxic RBCs. This finding suggests that not only increased erythrocyte number, but also cell-intrinsic phenotypic changes in hypoxic RBCs, contribute to glucose lowering. Radioactive tracing experiments using carbon-13–labeled 2-deoxy-D-glucose (U-¹³C 2DG) revealed increased glucose uptake by hypoxic erythrocytes. This was corroborated by elevated expression of glucose transporters GLUT1 and GLUT4. The absorbed glucose was preferentially funneled into glycolysis under hypoxic conditions.
“ ” —
” —
Fig. 3 Glucose reduction is an erythrocyte intrinsic mechanisms via glucose transporter upregulation —
Finally, the authors explored the translational implications of hypoxia-induced erythrocyte glucose uptake. First, they examined whether hypoxia could ameliorate hyperglycemia in streptozotocin (STZ)-induced diabetic mice. Hypoxia exposure led to erythrocytosis, reduced blood glucose levels, and improved glucose tolerance in STZ-treated animals. Second, in a cell therapy–like approach, transfusion of hypoxic RBCs into STZ-induced diabetic mice attenuated hyperglycemia. Lastly, the authors tested a small molecule, HypoxyStat, which reduces oxygen offloading into tissues and thereby induces localized tissue hypoxia. Treatment with HypoxyStat reduced hyperglycemia in mice fed a high-fat diet (HFD).
“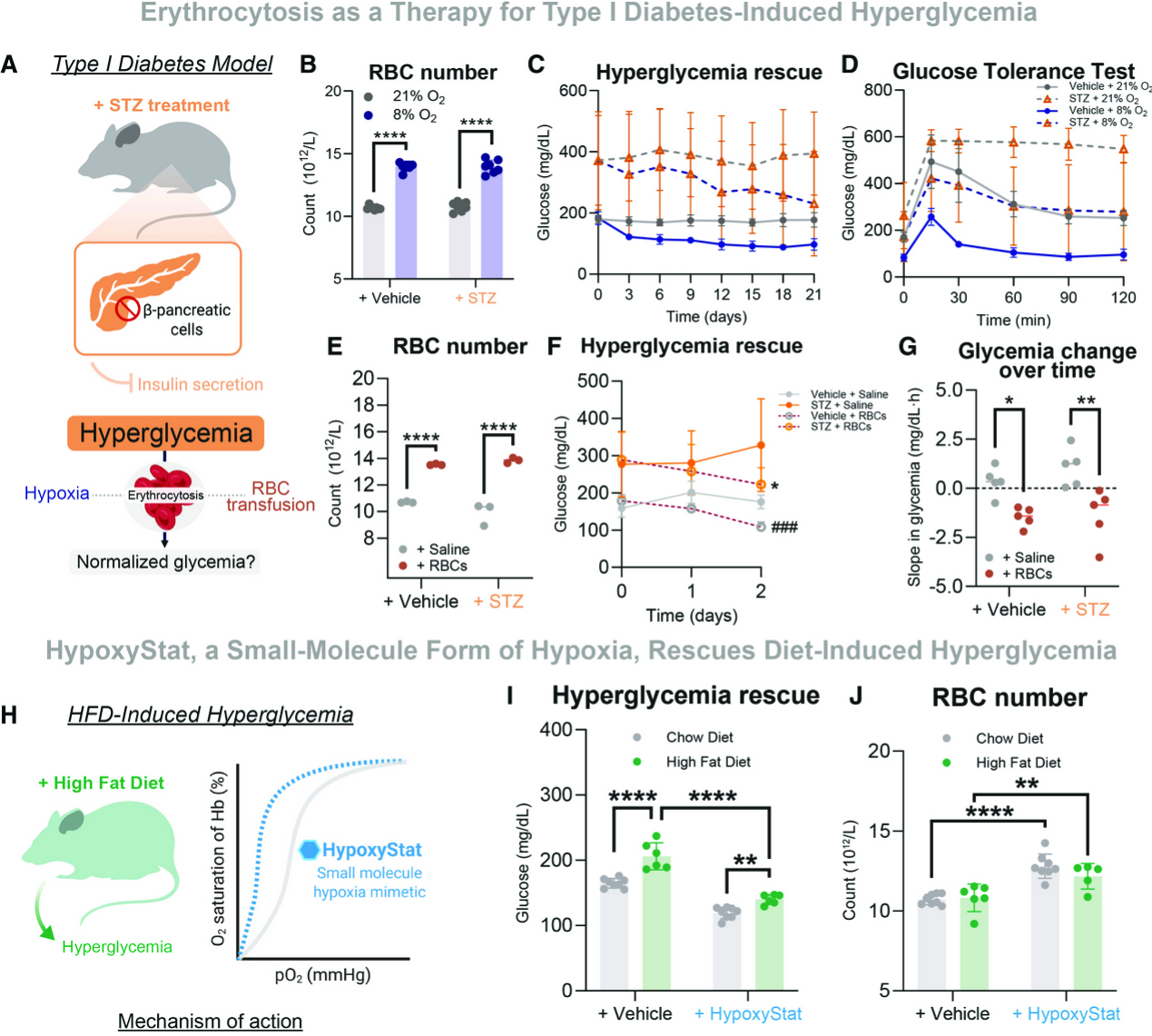 ” —
” —
Fig. 4 Preclinical model to test translational application of hypoxia-induced erythrocytosis in diabetes —
Conclussion and Study Limitations — Overall, this study establishes the importance of hypoxia-induced erythrocytosis in lowering blood glucose levels, providing a mechanistic explanation for the association between high-altitude living and reduced diabetes incidence. Nevertheless, several limitations should be considered. The experiments were conducted exclusively in the C57BL/6J strain, which possesses distinct metabolic characteristics that may influence glucose homeostasis. Validation in additional mouse strains will therefore be essential to determine the broader applicability of these findings.
Aging was not examined in this study, despite its well-established association with reduced erythropoiesis and increased diabetes prevalence. It will be important to assess whether the hypoxia–erythrocyte–glucose axis remains effective in older organisms. Furthermore, the molecular mechanisms underlying the increased expression of glucose transporters in erythrocytes generated under hypoxic conditions were not explored. These changes may originate at the level of erythroid progenitor cells or even upstream hematopoietic stem and progenitor cells, suggesting a developmental or epigenetic component to the observed phenotype. Importantly, the role of hypoxia-inducible factor 1 alpha (HIF-1α), a central regulator of cellular responses to low oxygen, was not addressed and warrants future investigation using HIF-1α knockout and knock-in mouse models.
Although multiple experimental approaches in this study highlight the therapeutic potential of leveraging hypoxia to ameliorate hyperglycemia, the possible adverse effects of hypoxia pathway activation were not addressed. Chronic or systemic hypoxia can exacerbate pathological conditions such as cancer and chronic inflammation, both of which are known to thrive in hypoxic microenvironments. Careful evaluation of safety, dosing, and context-specific effects will therefore be critical before considering hypoxia-based strategies for diabetes treatment.
